Avian Tuberculosis is a chronic wasting condition affecting many species of birds. This is not a disease that I have seen much in commercial poultry. I have learnt of one case in racing pigeons recently, seen a case in hens at a processing plant in the late 1980’s and know of cases in avian collections and rescued hens on free range. You would expect to see the disease in situations where birds live for many years on the same ground and in environments that cannot be easily cleaned.
Table of Contents
Synonyms
Avian TB
Cause
Mycobacterium avium. Classification is complex. The 4 subtypes are:-
| Species | Serotype | Host |
| M.avium subsp avium | 1,2,3 | virulent for birds + pigs, rabbits, mink |
| M.avium subsp hominissuis | 4-6, 8-11, 21 | external environment, dust, water, soil – invertebrates and some virulent for birds |
| M.avium subsp paratuberculosis | ruminants and other animals | |
| M.avium subsp silvaticum | rare, virulent for birds |
M.avium subsp avium is an acid fast organism when stained by the Ziehl-Neelsen method. They are highly resistant organisms that can survive in the environment for years. Viable organisms have been recovered from litter and soil 4 years after removal from an infected flock. It has also been shown to survive in sawdust for 168 and 244 days at 20oC and 37oC respectively.
Host
Many Avian species – chickens>ducks>geese>turkeys and also pheasants, birds in zoological collections and cage birds. Pigs and rabbits are also susceptible. Man – infection rare as man considered to be very resistant but it can occur after immunosuppressive disease.)
Transmission
Close contact with the live or dead bird, ingestion of faeces or faecal aerosols, contaminated ground and equipment. Bacteria have been found in eggs but there is no evidence to suggest that chicks hatched from infected eggs develop the disease. Eggs are unlikely to be the route of infection for a flock.
Clinical signs
The disease has a long incubation period. Therefore, it is usually seen in birds 12 months or older. Loss of condition with birds taking weeks to months to die. Continual low mortality and lost egg production. Appetite may remain good despite cachexia. Disease is commoner in extensive systems and zoos where birds are kept for more than a year. the skin may be pale and comb may be small shrunken. Lameness can be associated with bone infection. Diarrhoea with soiling of the vent feathers can be indicative of intestinal lesions. No specific egg abnormalities but the hen will stop laying.
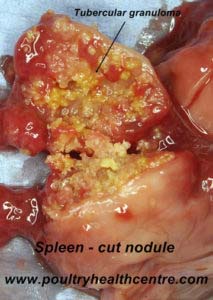
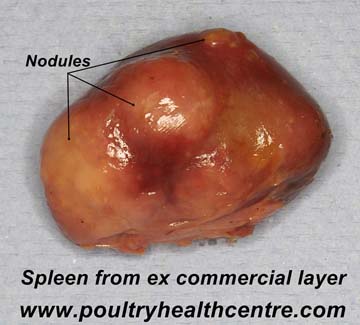
Pathology
Very poor condition with tubercular granulomata which are variable sized, irregular grey to white nodules that can be found in the liver, spleen, intestine and bone marrow. When cut the granulomas are firm, with pale yellow caseous centres, sometimes bile stained in the liver and intestine. Confirmation of the diagnosis can be made by staining impression smears with Ziehl-Neelsen method.
Diagnosis
the history, clinical signs and post mortem lesions are suggestive of the diagnosis. Confirmation of the species of acid fast bacteria should be undertaken by culture or genetic identification. The tuberculin test is available for poultry with avian tuberculin (0.05 – 0.1ml) being injected into one wattle (use a 1cm, 25g needle) and 48 hours later the prescence of swelling and heat being compared with the uninocculated wattle. This test will detect about 80% of positive birds and can miss birds with severe lesions. A stained antigen, whole blood agglutination test and an ELISA test are also available. As none of these tests will detect 100% of infected birds they will need to be used in planned monitoring programme.
Differential diagnoses
Marek’s disease, impactions, coccidiosis, other tumours, other coause of lameness
Treatment
There is no specific treatment and avian mycobacteria are particularly antibiotic resistant.
Prevention
Control is by testing birds – PCR or Z-N staining of faeces, tuberculin test (wattle in the chicken), agglutination and ELISA tests. Remove identified carriers. Good hygiene and management and the placing unifected stock on clean groher concentration than that used for disinfecting aganst NDV or AI.
Erysipelas is the 17th commonest diagnosis made by my local AHVLA labs. Details of species affected, clinical signs and post mortem lesions observed can be found by clicking here.
